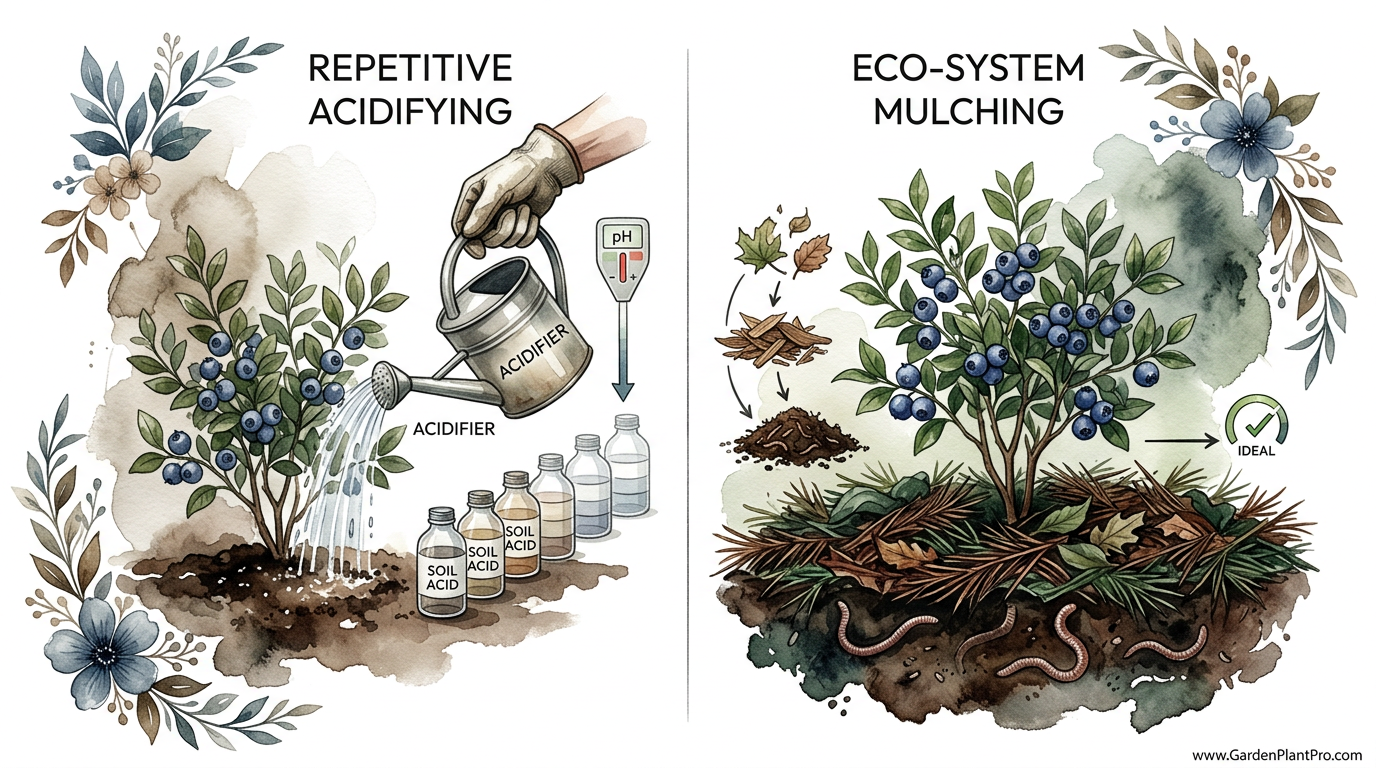
Most gardeners spend hundreds on soil amendments when the permanent solution is actually free under your local trees. I watched my neighbor spend $200 a year on soil acidifiers while my blueberries outperformed hers for $0. The secret isn’t in a bag; it’s in the mulch. By mimicking the forest floor, you create a self-sustaining acid battery that feeds your berries for years.
Most people treat blueberries like a typical vegetable garden, but these plants are actually forest edge dwellers. They evolved to live in the damp, acidic, and fungal-rich debris of the woodland floor. When we try to force them into regular garden soil, we are essentially asking a fish to live in a desert. Understanding how to bridge that gap naturally is the key to baskets full of sweet, blue fruit every summer.
How To Acidify Blueberry Soil Naturally
Blueberries are part of the Ericaceae family, which makes them cousins to azaleas and rhododendrons. These plants are unique because they thrive in soil with a pH between 4.5 and 5.5. In this narrow range, essential nutrients like iron and manganese become chemically available for the plant to drink up. If the pH rises even slightly above 6.0, the plant begins to starve, regardless of how much fertilizer you dump on it.
Natural acidification involves two distinct paths. One is the immediate physical change brought by adding acidic organic matter like sphagnum peat moss. The other is the long-term biological change driven by soil bacteria and the slow breakdown of wood-based mulches. Creating a forest-like environment relies on building a high-carbon, fungal-dominated soil structure that naturally keeps the pH low over the lifespan of the bush.
Gardeners usually encounter high pH issues because of the parent material of their soil or the alkalinity of their local water supply. Natural methods focus on working with the local ecosystem rather than fighting it. Instead of reaching for synthetic chemicals that provide a short-term spike in acidity, we look at materials like pine needles, elemental sulfur, and aged sawdust. These materials act like a slow-release battery, constantly refreshing the acidity as they decay.
Building the Underground Acid Battery
The most effective way to drop soil pH naturally over time is through the use of elemental sulfur. This is a naturally occurring mineral, often found near volcanic vents, and it is the gold standard for organic growers. It does not work by a simple chemical reaction. Instead, it relies on specialized soil bacteria called Thiobacillus. These tiny microbes eat the sulfur and, through their natural metabolic process, excrete sulfuric acid into the soil.
Because this is a biological process, it requires specific conditions to work. The soil must be moist, and temperatures need to be above 55°F (13°C) for the bacteria to wake up and start “cooking.” If you apply sulfur in the dead of winter, nothing will happen until the spring thaw. This slow-moving nature is actually a benefit because it prevents the “pH yo-yo” effect that can stress out sensitive blueberry roots.
Another heavy hitter in the natural arsenal is sphagnum peat moss. It has a natural pH around 4.0 to 4.5. When you dig a planting hole, mixing in equal parts peat moss and native soil provides an immediate “acid pocket” for the young roots. While peat is a fantastic starter, it eventually breaks down and disappears. This is why you must pair it with a long-term mulching strategy to maintain those levels for the next twenty years of the plant’s life.
The Forest Floor Strategy: Mulch as Medicine
Pine needles are often surrounded by myths in the gardening world. Many people claim they don’t actually lower pH, while others say they are the only thing that works. The truth lies in the middle. Fresh pine needles have a pH of about 3.2 to 3.8. While they won’t drop a pH of 7.5 down to 5.0 on their own, they are incredible at maintaining an already acidic environment.
A thick layer of pine straw, about 4 to 6 inches (10 to 15 cm) deep, creates a protective blanket. This blanket prevents alkaline rain runoff from neutralizing the soil and provides a steady, slow drip of organic acids as the bottom layer of needles decomposes. Oak leaves are another fantastic resource. While green oak leaves are somewhat neutral, shredded brown oak leaves provide the high-carbon environment that helps keep the soil acidic and fungal-active.
Wood chips from coniferous trees, such as pine, spruce, or cedar, are superior to hardwood chips for blueberries. Hardwood chips from trees like maple or ash can sometimes have a more neutral or even slightly alkaline effect as they rot. Conifer wood, however, stays acidic for much longer. These chips also encourage the growth of ericoid mycorrhizal fungi. These specific fungi form a partnership with blueberry roots, helping them scavenge for nutrients in the low-fertility, high-acid environments they love.
Managing Your Water Source
One of the biggest hidden killers of soil acidity is the garden hose. Most municipal tap water is treated to be slightly alkaline, often with a pH of 8.0 or higher, to prevent pipe corrosion. If you water your blueberries with city water every day during a dry summer, you are essentially “liming” your soil with every drop. Over a few months, this can completely neutralize all the hard work you did with sulfur and peat.
Rainwater is naturally acidic, usually hovering around a pH of 5.6. Collecting rain in barrels is the single best way to maintain your blueberries naturally. If you must use tap water, you can neutralize its alkalinity by adding a small amount of white vinegar or citric acid to the watering can. About 1 to 2 tablespoons (15 to 30 ml) of vinegar per gallon (3.8 liters) of water is usually enough to take the edge off the alkalinity without harming the soil life.
Benefits of Natural Acidification
Choosing natural methods over synthetic ones creates a more resilient plant. Natural amendments improve soil structure and water retention. Because blueberries have very shallow, fibrous roots that lack traditional root hairs, they are extremely sensitive to drought. A thick, natural mulch layer keeps the root zone cool and moist even in the heat of July.
- Nutrient Availability: In acidic soil, iron, phosphorus, and potassium are easily absorbed.
- Microbial Health: Organic amendments foster a diverse ecosystem of fungi and bacteria that protect the plant from root rot.
- Cost Efficiency: Materials like pine needles and autumn leaves are free, reducing the need for expensive bagged fertilizers.
- Flavor Density: Berries grown in soil rich in organic matter and natural minerals often have a deeper, more complex flavor than those grown with synthetic salts.
Common Mistakes to Avoid
The most frequent error is applying too much sulfur at once. If you dump massive amounts of sulfur onto the soil, the bacteria may produce acid faster than the soil can buffer it, leading to a “hot spot” that can burn the delicate blueberry roots. It is always better to apply smaller amounts twice a year—once in early spring and once in early autumn—to let the soil adjust gradually.
Another mistake is using the wrong type of compost. Standard kitchen compost often contains eggshells or wood ash, both of which are high in calcium and will raise the soil pH. For blueberries, you want an “acid compost” made primarily of pine needles, oak leaves, and coffee grounds. Avoid using mushroom compost or composted manure, as these are typically too “sweet” (alkaline) and salty for blueberries to handle.
Comparing Natural Acidifiers
| Amendment | Speed of Action | Longevity | Primary Benefit |
|---|---|---|---|
| Elemental Sulfur | Slow (3-12 months) | Very High (Years) | Permanent pH reduction through biology. |
| Sphagnum Peat Moss | Immediate | Medium (1-3 years) | Creates an instant acid pocket for new roots. |
| Pine Needle Mulch | Very Slow | High (Ongoing) | Maintains acidity and regulates temperature. |
| Vinegar/Citric Acid | Instant | Very Low (Days) | Neutralizes alkaline irrigation water. |
| Ammonium Sulfate | Fast | Low (Months) | Provides nitrogen while lowering pH slightly. |
Practical Tips for Success
Start by getting a professional soil test. You cannot accurately guess your soil’s pH by looking at it. Once you have your baseline, you can calculate exactly how much elemental sulfur you need. For sandy soils, you usually need about 1 pound (450 g) per 100 square feet to drop the pH by one point. For heavy clay, you might need 2 to 3 pounds (900 to 1350 g) for the same result.
Keep the mulch away from the actual stem of the bush. While blueberries love a thick blanket, piling mulch directly against the wood can trap moisture and cause crown rot. Leave a small 2-inch (5 cm) gap around the base of the plant to allow for air circulation. This ensures the roots stay happy while the “acid battery” works its magic just a few inches away.
Monitor your plants for signs of iron chlorosis. If you see the leaves turning yellow while the veins stay bright green, your soil pH is likely rising too high. This is the plant’s way of saying it can’t reach the iron in the soil. Before you reach for an iron spray, check the pH. Usually, fixing the acidity will unlock the iron that is already present in your dirt.
Advanced Considerations for the Serious Grower
Experienced growers often focus on the fungal-to-bacterial ratio of their soil. Most vegetable gardens are bacterial-heavy, which naturally trends toward a neutral pH. To keep blueberries happy for decades, you want to shift that balance toward fungi. Using “ramial” wood chips—chips made from small branches less than 3 inches (7.5 cm) in diameter—provides the perfect food source for the beneficial fungi that maintain acidic conditions.
DIY PROJECT: Collect rainwater no matter where you live...
This DIY project is the best way to legally collect rainwater NO MATTER where you live. Get chlorine-free water, cut down on your water bills, and have enough for an emergency situation or to water your garden. Read More Here...
Regional climate also plays a huge role. In very rainy areas, the soil tends to become acidic naturally as calcium leaches away. In arid regions, the lack of rain means minerals stay in the soil, keeping the pH high. If you live in a desert or high-plains environment, you may find it nearly impossible to keep the ground acidic enough. In these cases, growing blueberries in large, 20-gallon (75-liter) containers filled with a mix of peat moss and pine bark is often a more successful strategy than fighting the native earth.
Real-World Example: The Three-Year Turnaround
Consider a gardener in a suburban area with a native soil pH of 7.0. In the first year, they dig a wide, shallow trench, filling it with 50% peat moss and 50% native soil. They add the recommended dose of elemental sulfur across the entire surface and scratch it into the top 2 inches (5 cm). A 4-inch (10 cm) layer of pine straw is applied over the top.
By the second year, the soil test shows the pH has dropped to 6.2. The sulfur-eating bacteria are in full swing. The gardener adds another light application of sulfur and switches to watering exclusively with captured rainwater. In the third year, the pH hits the “sweet spot” of 5.0. The bushes, which were previously stunted and pale, suddenly explode with 3 feet (1 meter) of new growth and a heavy set of fruit buds. At this point, the system is self-sustaining; all that is needed is an annual top-dressing of fresh pine needles.
Final Thoughts
Lowering your soil pH naturally is a marathon, not a sprint. It requires patience and a shift in how you view the “dirt” in your yard. By mimicking the deep, dark, acidic floor of an old-growth forest, you provide your blueberry bushes with exactly what they evolved to crave. The results aren’t just seen in the health of the plants, but in the sheer abundance of the harvest.
Once you establish your “acid battery,” the maintenance becomes a simple seasonal rhythm of mulching and monitoring. You will save money, improve your local soil ecology, and enjoy berries that are far superior to anything found on a grocery store shelf. Experiment with the materials you have available locally, keep an eye on your water source, and let the soil biology do the heavy lifting for you.